A sodium atom has an atomic radius of about 2.23 angstroms.

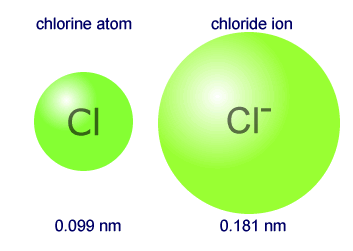
Therefore the atomic volume of sodium is 11.763. We expect POSITIVE ions to be SMALLER than the corresponding atom because there are fewer electrons than protons => an excess of POSITIVE charge => the electron cloud is more attracted to the nucleus on average and becomes more 'concentrated' => $\ce$ carries no unbalanced charge and has comparable valence principal quantum number to the ions, so we expect its radius to fall somewhere between the positive ions (which are smaller) and the negative ions (which are larger). Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of sodium-23 (atomic number. On the periodic table, atomic radius generally decreases as you move from left to right across a period (due to increasing nuclear charge) and increases as you move down a group (due to the increasing number of electron shells). Because of having low atomic mass and large atomic radius, sodium is one of the least dense elements (third least dense element first two are lithium and.The ionic radius for Na+ is 102 pm and for Cl is 181 pm. if the atomic radius of sodium atoms is 1.86 10 -10 m how many sodium atoms are needed to make a line that is 1.00 CM in length Answer Discussion Video. A periodic table coupled with simple electrostatic arguments should suffice to provide at least a rough ordering of ions and atoms. The atomic radius of sodium is 186 pm and of chlorine is 100 pm.
